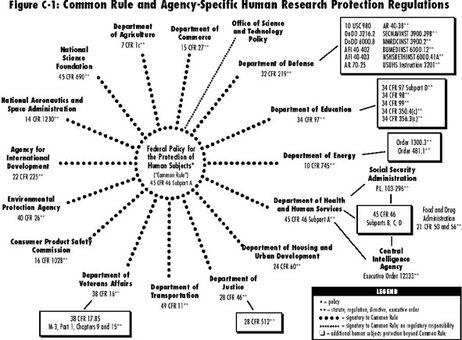
Title 45, Part 46 of the Code of the Federal Regulations in Protection of Human Subjects
|
i. The Common Rule is a regulatory framework that applies to 17 federal agencies that provides funding for research on human subjects. It essentially provides guidelines to conducting these types of research and a failure to abide by their stipulations warrants potential sanctions, suspensions or termination of the research project (Gostin, Vanchieri, Pope 76).
ii. The Common Rule provides various types of protection mechanisms for human subjects, such as IRB (Institutional Review Board) review process for research and Informed consent processes. iii. Concerning IRB process, the Common Rule states that, “when some or all of the subjects are likely to be vulnerable to coercion or undue influence, additional safeguards have been included to protect their rights and welfare of those subjects” (Gostin, Vanchieri, Pope 77). iv. With informed consent, the disclosure requirements are specified below: 1. A description of the study and its intended purposes 2. An identification of any potential risks to the research subject 3. A description of any benefits that could be gained 4. A disclosure of alternative treatments that could be beneficial 5. A description of how confidentiality will be maintained 6. With minimal risk experiments, an explanation of the potential consequences from participating in the experiment. 7. Contact information for potential questions or concerns 8. A statement describing that participation is voluntary and a loss of refusal will not result in a penalty In the Common Rule, additional protection is also provided for vulnerable populations like pregnant women, fetuses and neonates (Subpart B), prisoners (Subpart C), and children (Subpart D). |